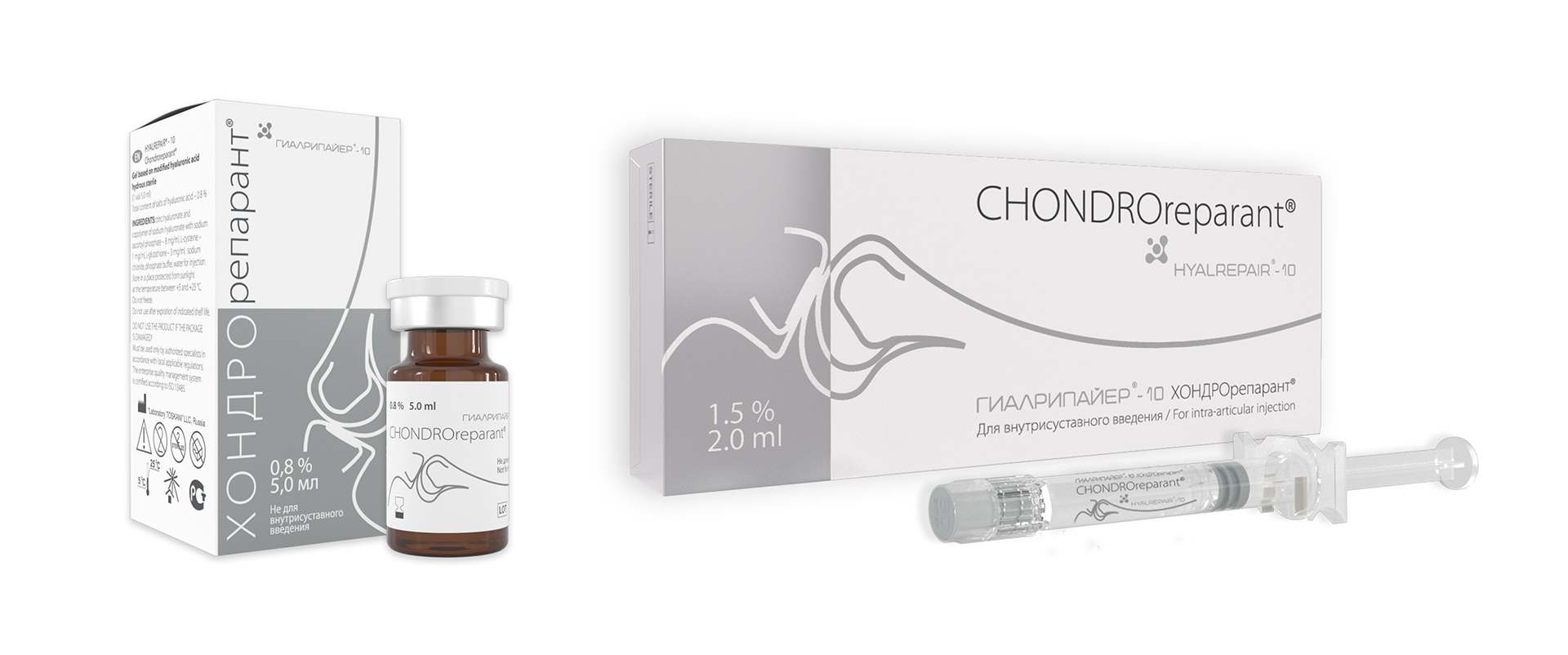
Chondroreparants
are an unique injectable line of medical devices for innovational non-medicinal preventive health care for people who lead an active lifestyle and for the treatment of different pathology of the locomotor system.
ACTION
- The line of injectable medical devices for application in traumatology and orthopedics, rheumatology, neurology, sports medicine and for sustaining prophylaxis during extreme physical activities.
Unique series of formulas with targeted action are based on stabilizes hyaluronic acid with proteinogenic amino acids and antioxidant complex. - Immediate and delayed effects based on free fraction which provides fast start of antioxidant and reparative processes and stabilized fraction that supplies deferred effect due to the prolonged biodegradation and gradual release of active components.
- Formulas were created specially for compensation of deficiency of hyaluronic acid and active regulating molecules and for progressing clinical effects such as reparation of the connective tissue structure or cell oxidative stress inactivation.

 |
 |
|
| PRODUCT | CHONDROREPARANT® HYALREPAIR®-02 | CHONDROREPARANT® HYALREPAIR®-10 |
| Ingredients | Hyaluronic Acid modified with ascorbyl phosphate, proline, glycine, lysine | Hyaluronic Acid modified with ascorbyl phosphate, cysteine, glutathione |
| Action | structural proteins synthesis stimulation | blocking free radical destruction of cells |
| Indications | degenerative-dystrophic connective tissue alterations (joint cartilage, synovial membrane, periarticular structures etc.) | post-inflammatory connective tissue alterations (joint cartilage, synovial membrane, periarticular structures etc.) |
| Dosage | 2.0 ml (syringe) 5.0 ml (vial) |
2.0 ml (syringe) 5.0 ml (vial) |












